Funny stainless steel fastener companies reply to my question
#17
Nordschleife Master
#18
Rest in Peace
Rennlist Member
Rennlist Member
Join Date: May 2006
Location: Bird lover in Sharpsburg
Posts: 9,903
Likes: 0
Received 2 Likes
on
2 Posts
Good for you!! Nobody said it couldn't be done. Friend of mine has a big block Olds Toranado drivetrain in a 50's Beetle.
No matter how you slice it or justify it, still going to be considered a hack job by most. If you don't care, then go ahead.
Just like some people who feel it's ok to cut holes in the fender to make clutch jobs easier.
No matter how you slice it or justify it, still going to be considered a hack job by most. If you don't care, then go ahead.

Just like some people who feel it's ok to cut holes in the fender to make clutch jobs easier.
#19
Administrator - "Tyson"
Lifetime Rennlist
Member
Lifetime Rennlist
Member
#20
Rennlist Member
I don't know much, but owning a sailboat does encourage one to learn something about corrosion. Any two different metals will form a battery and corrode, what's missing here is any sort of electrolyte.
Any conductive material has a natural galvanic potential (electrical voltage, when made part of a battery). Metals with similar potential won't corrode in combination. Here's a table of galvanic potential from corrosionist.com:
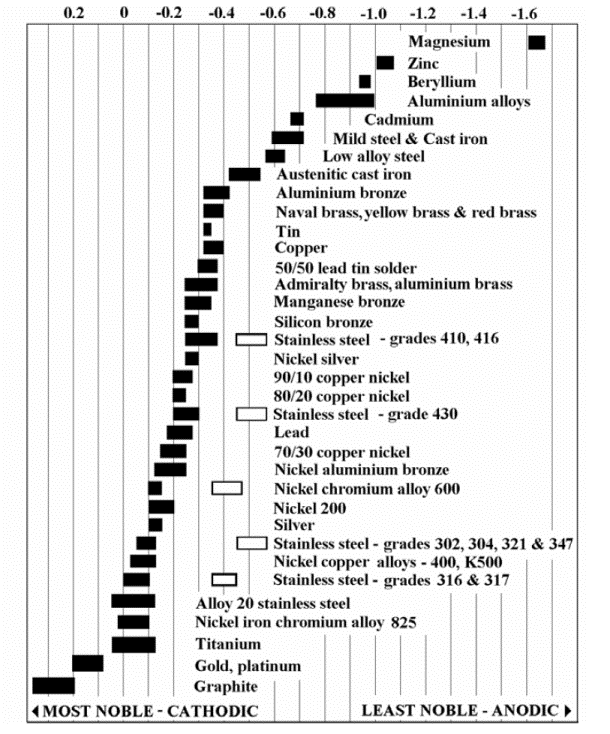
Electrical potential is the horizontal scale-- metals close together horizontally have a smaller difference and make a lousy battery. Note zinc in the upper-right and graphite (carbon) in the lower-left-- a common drycell battery. The material that is more anodic-- farther to the right-- corrodes and gets converted to something else (the ubiquitous white powder), while the material to the left is protected. Zinc is used on boats to protect other metals, and if you get magnesium anywhere near a boat it just goes away.
Cadmium, steel, aluminum are all pretty close together. Stainless is all over the place depending on alloy (most fasteners are 318 I think).
But it takes an electrolyte to make the battery work, nothing will happen if it is dry with no conductive path to complete the electrical circuit. Salt water makes a terrific electrolyte, but the 928 exhaust studs are pretty well protected against road stray (assuming belly pans). Suspension parts, not so much.
So cad-plated steel is better than stainless, but in any event the guy was right-- his stainless studs are not at risk. If there is any corrosion, it will be the aluminum block. And that is only an issue if conductive (salty) water gets to it.
Zinc plating is theoretically better at protecting steel bolts than cad plating, the problem is that the zinc plating is thin and goes away quickly in the process of protecting the steel. But it's what we've got.
Any conductive material has a natural galvanic potential (electrical voltage, when made part of a battery). Metals with similar potential won't corrode in combination. Here's a table of galvanic potential from corrosionist.com:

Electrical potential is the horizontal scale-- metals close together horizontally have a smaller difference and make a lousy battery. Note zinc in the upper-right and graphite (carbon) in the lower-left-- a common drycell battery. The material that is more anodic-- farther to the right-- corrodes and gets converted to something else (the ubiquitous white powder), while the material to the left is protected. Zinc is used on boats to protect other metals, and if you get magnesium anywhere near a boat it just goes away.
Cadmium, steel, aluminum are all pretty close together. Stainless is all over the place depending on alloy (most fasteners are 318 I think).
But it takes an electrolyte to make the battery work, nothing will happen if it is dry with no conductive path to complete the electrical circuit. Salt water makes a terrific electrolyte, but the 928 exhaust studs are pretty well protected against road stray (assuming belly pans). Suspension parts, not so much.
So cad-plated steel is better than stainless, but in any event the guy was right-- his stainless studs are not at risk. If there is any corrosion, it will be the aluminum block. And that is only an issue if conductive (salty) water gets to it.
Zinc plating is theoretically better at protecting steel bolts than cad plating, the problem is that the zinc plating is thin and goes away quickly in the process of protecting the steel. But it's what we've got.
#21
Captain Obvious
Super User
Super User
Never mind the galvanic reaction. Stainless is usually really soft and shouldn't be used in exhaust satuds or collector bolts.
#23
Three Wheelin'
Join Date: Feb 2009
Location: Washington "Dc"
Posts: 1,810
Likes: 0
Received 0 Likes
on
0 Posts
What about good ole anti-seize on the bolts (Smear) as they are installed. I've always used it on header studs/bolts and never had a problem with them becoming stuck from corrosion.
#24
Rennlist Member
Second pull and install was done in less than 8 hours. Seems like 2 of them were dealing with the damn wiring harness through the firewall.
#26
Three Wheelin'
Thread Starter
You learn something new everyday, thanks for the insight on fasteners, especially jcorenman, that was very interesting. I've also heard of engine blocks corroding if the pH is at the correct level to form an electrolyte and thus a large battery that, like you said, corrodes the anodic material (would this be the negative as the positive or less anodic material draws electrons from the anodic material that wish to be positively charged?).
#27
Not the sharpest tool in the shed
Rennlist Member
Rennlist Member
#28
Rennlist Member
Jeff, how the heck did you get the engine between the motor mounts without an extra set of hands? Mine were new from Roger. They were very tight. I thought I was going to have to install it myself but my son showed up for a visit just in time...However, it was a b%#*&ch even with help.
Man, that must have been some day,
Man, that must have been some day,
#29
Captain Obvious
Super User
Super User
Jeff, how the heck did you get the engine between the motor mounts without an extra set of hands? Mine were new from Roger. They were very tight. I thought I was going to have to install it myself but my son showed up for a visit just in time...However, it was a b%#*&ch even with help.
Man, that must have been some day,
Man, that must have been some day,
#30
Nordschleife Master
I'm guessing you guys are talking about different motor mounts. The old style are tough to get past and those heat shields don't cooperate well, either.


