06 C2S Tire/air Pressure
#1
Hello everyone,
I have a bad back and need a comfortable ride and am seeking advice on tire/air pressure for my Michelin Pilot SS's:
235/35 ZR 19
295/30 ZR 19
would like to run the pressures on the low side, but safely.
Any and all advice appreciated.
Thanks,
John
I have a bad back and need a comfortable ride and am seeking advice on tire/air pressure for my Michelin Pilot SS's:
235/35 ZR 19
295/30 ZR 19
would like to run the pressures on the low side, but safely.
Any and all advice appreciated.
Thanks,
John
#2
Rennlist Member
I ran 33/37 cold PSI on mine.
#3
Nordschleife Master
#5
RL Community Team
Rennlist Member
Rennlist Member
If you can find a place that uses Nitrogen for your tire fill, it does not expand and contract nearly as much as regular air so your pressures will stay more consistent even if measured when cold outside or after a long drive on a high temperature day. Racers use it to minimize that variable for their suspension tuning, but in your case seems it might help keep a compliant ride at close to a set pressure for your back. Pressures can be fine tuned with regular air so you don't need to keep going back to the shop; it will just slightly dilute the Nitrogen concentration. Another advantage is that the molecule size is larger so it doesn't slowly seap out through the rubber tire like air does.
#7
Rennlist Member
Trending Topics
#8
Thank you all, much appreciated and will heed advice! Nitrogen sounds like a practical solution for now. Yes, I've considered 18's wheels, but as the car is new to me, I'm just in the process of sorting it out and it currently has new Michelins on these 19" rims.
#9
Nordschleife Master
The only advantage of Nitrogen (N2) is that the N2 source is moisture-free. Nitrogen compresses and expands exactly as air. Moisture-free N2 may be useful if you live in an area of high humidity and regular air contains a significant amount of water vapor. However, filling with N2 only avoids moisture if the moisture is fully extracted from the tire prior to filling with N2, which requires a special machine and tortures tires.
And of course, to maintain the N2 moist-free advantage requires finding a N2 filling station on the road wherever you are.
The theory that N2 has a larger molecular size... regular air already is 80% N2 and the molecular size argument is a marketing ploy.
And of course, to maintain the N2 moist-free advantage requires finding a N2 filling station on the road wherever you are.
The theory that N2 has a larger molecular size... regular air already is 80% N2 and the molecular size argument is a marketing ploy.
#10
Nordschleife Master
#11
Nordschleife Master
It always surprises me to read posts complaining about the car's suspension stiffness. If one considers a PASM equipped car stiff, one has never driven/been in a stiff suspension car. The 997 has a pretty cushy ride. Besides... these are sports cars.
#12
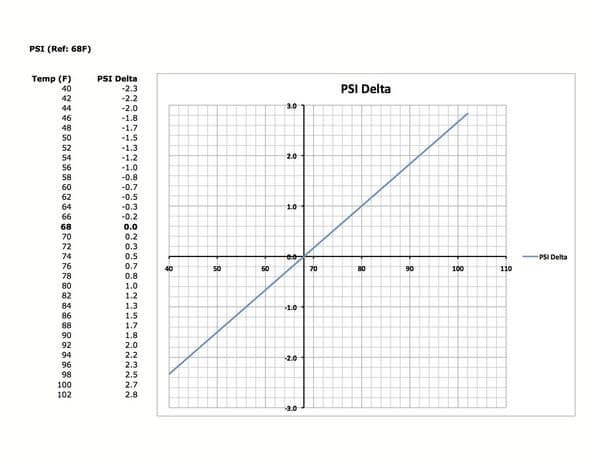
I would recommend about 36 cold, and dont forget, if you dont have the right pressure you are going to wear your tires more prematurely. Secondly, I agree Nitro has only one advantage, no moisture (for the TPMS sensors sake) My experience, left on a trip from Fla (75 degree) with 42-44psi hot tires,and went to freezing temp in NC (40-25degree) Woke up with pire pressure warning, 25psi, drove few miles and rose to 34, then 38. Finished and returned home, never added any air and still running 43-44psi.
#13
RL Community Team
Rennlist Member
Rennlist Member
The only advantage of Nitrogen (N2) is that the N2 source is moisture-free. Nitrogen compresses and expands exactly as air. Moisture-free N2 may be useful if you live in an area of high humidity and regular air contains a significant amount of water vapor. However, filling with N2 only avoids moisture if the moisture is fully extracted from the tire prior to filling with N2, which requires a special machine and tortures tires.
And of course, to maintain the N2 moist-free advantage requires finding a N2 filling station on the road wherever you are.
The theory that N2 has a larger molecular size... regular air already is 80% N2 and the molecular size argument is a marketing ploy.
And of course, to maintain the N2 moist-free advantage requires finding a N2 filling station on the road wherever you are.
The theory that N2 has a larger molecular size... regular air already is 80% N2 and the molecular size argument is a marketing ploy.
http://www.popularmechanics.com/cars...a3894/4302788/
#14
Nordschleife Master
According to Popular Mechanics, what I stated about Nitrogen is correct.
http://www.popularmechanics.com/cars...a3894/4302788/
http://www.popularmechanics.com/cars...a3894/4302788/

#15
I'm with you. It's that pesky PV=nRT thing. This guy has a pretty good explanation and some data to show N2 is BS.
http://www.barrystiretech.com/nitrogeninflation.html
http://www.barrystiretech.com/nitrogeninflation.html